 
Extensive washing will not cause a loss of precipitate, as the Ni(DMG) 2 complex is extremely insoluble (less than 1.0 mg per liter in cold water). The digested precipitate is filtered and washed extensively with distilled water to remove adsorbed impurities. Such simmering of a precipitate to encourage flocculation (a common technique) is known as digestion. A more tractable precipitate is generated if the solution is simmered for an hour or more to encourage the growth of larger crystals. The precipitate (Ni(DMG) 2 ) initially produced by adding DMG consists of very small crystals which cannot be filtered, as they penetrate or clog the filter mat. 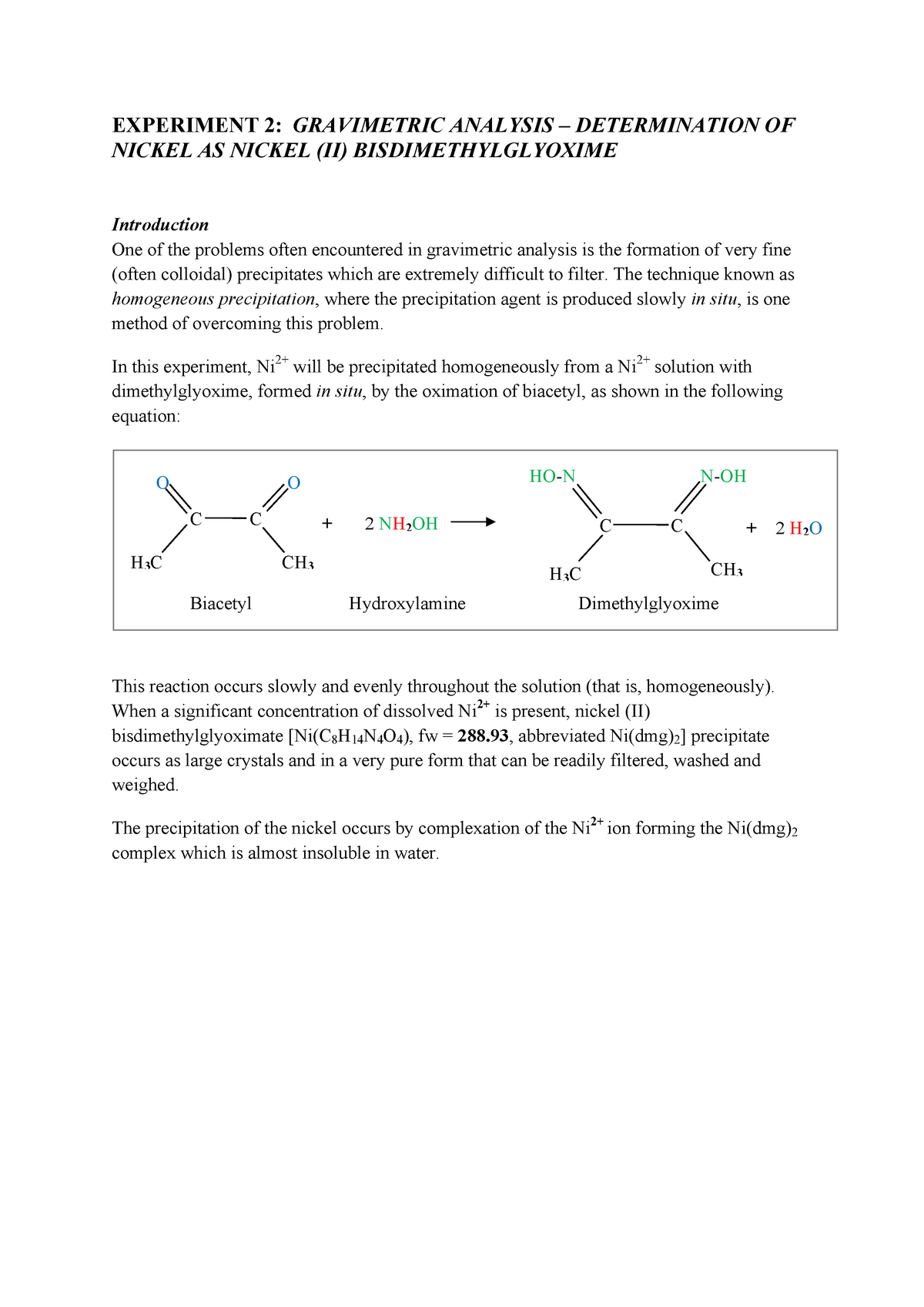
To avoid interference by Cu 2+, sodium tartrate is also added, as the tartrate ion wraps itself around the Cu 2+ ions and prevents their precipitation when the DMG is added. Before adding the DMG and separating the Ni 2+ from the Cu 2+ ions, the solution must be neutralized (and made weakly basic) by adding sodium acetate. The resulting solution is acidic and contains both Ni 2+ and Cu 2+ ions. Reaction of the coin with the nitric acid oxidizes both the nickel and copper atoms and releases various oxides of nitrogen:ģ Cu (s) + 8 H + (aq) + 2 NO 3 - (aq) -> 3 Cu 2+ (aq) + 2 NO (g) + 4 H 2O (l) ģ Ni (s) + 8 H + (aq) + 2 NO 3 - (aq) -> 3 Ni 2+ (aq) + 2 NO (g) + 4 H 2O (l) Before the five-cent piece can be analyzed for the nickel content, it must be cleaned, weighed and then dissolved. The Ni(DMG) 2 complex (MW 288.9 g/mole) is stable to heat and is unusually hydrophobic it will dry quickly in an oven at 110-120 oC to yield a pure substance whose weight can be used to calculate the nickel content of the original sample.Ī five-cent piece (a nickel) consists of copper alloyed with nickel and other metals. The reaction is illustrated below the dashed lines represent hydrogen bonds which help stabilize the complex.Ģ Dimethylglyoxime + Ni 2+ -> Ni(DMG) 2 + 2 H + No other common metallic ion reacts in the same way, so the formation of the insoluble Ni(DMG) 2 complex makes it possible to separate the Ni 2+ ions from other metal ions in solution. In the case of nickel, Ni 2+ ions form an extremely insoluble bright red complex with two dimethylglyoxime (DMG) anions under mildly basic conditions. That usually means that a material dissolved in solution is completely precipitated from solution, and the solid is weighed. Experiment 7-023: Gravimetric determination of Nickel in a Five-cent CoinĪ gravimetric determination of a substance involves the separation of that substance in a form which may be weighed accurately and is of known composition. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
